|
|||||||||||||
|
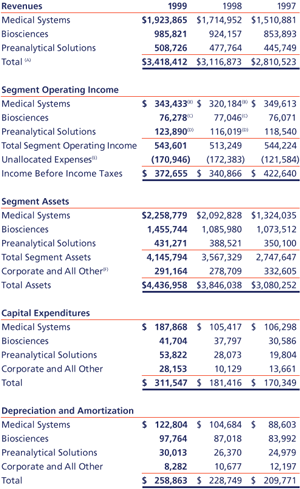
On September 30, 1999, the Company adopted the provisions of SFAS No. 131, “Disclosures about Segments of an Enterprise and Related Information.” SFAS No. 131 establishes a new method for the reporting of operating segment information based on the manner in which management organizes the segments within a company for making operating decisions and assessing performance. Segment data has been restated to conform to the SFAS No. 131 requirements for all periods presented. The Company’s organizational structure is based upon its three principal business segments: BD Medical Systems (“Medical”), BD Biosciences (“Biosciences”), and BD Preanalytical Solutions (“Preanalytical”). The Company’s segments are managed separately because each requires different technology and marketing strategies. The major products in the Medical segment are hypodermic products, specially designed devices for diabetes care, prefillable drug delivery systems, infusion therapy products, elastic support products and thermometers. The Medical segment also includes disposable scrubs, specialty needles, and surgical blades. The major products in the Biosciences segment are clinical and industrial microbiology products, flow cytometry systems for cellular analysis, tissue culture labware, hematology instruments, and other diagnostic systems, including immunodiagnostic test kits. The major products in the Preanalytical segment are sample collection products and specimen management systems. This segment also includes consulting services and customized, automated bar-code systems. The Company evaluates performance based upon operating income. Segment operating income represents revenues reduced by product costs and operating expenses. The calculations of segment operating income and assets are in accordance with the accounting policies described in Note 1. Distribution of products is both through distributors and directly to hospitals, laboratories and other end users. Sales to a distributor which supplies the Company’s products to many end users accounted for approximately 11% of revenues in 1999, 11% in 1998 and 10% of revenues in 1997, and were made from each of the Company’s segments. No other customer accounted for 10% or more of revenues in each of the three years presented.
|
|||||||||||||
|
|
|
|