|
||||
|
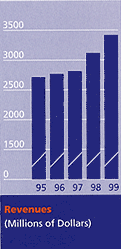
As a final point on fiscal 1999, I would observe that we are still in the early stages of a transformational journey toward accelerating our top line growth and increasing our clinical relevance. Thus, the only real mistake would be for us to let unwanted, but natural, growing pains deflect us from our chosen path. We can manage through the ebb and flow that goes with being a major medical technology company doing business all over the world. We are a leader whose products and markets are strong and vibrant. Because of that - and my faith in my associates throughout BD - I can reaffirm our commitment to our key long-term financial objectives: 15 percent earnings per share growth and accelerating revenue growth to 10 percent annually. I am confident that BD is superbly well positioned to realize both opportunities. Turning to accomplishments for the year, let me briefly summarize progress within the framework of our strategy for growth through acquisitions and alliances, new products, and geographic expansion. Acquisitions and alliances During fiscal 1999, BD completed several very important acquisitions. Going forward, we intend to remain alert to attractive opportunities, both strategically and opportunistically. The largest of our FY 99 acquisitions was completed in August when we closed on the purchase of Clontech Laboratories, Inc. This move significantly expanded our presence in the growing areas of molecular-based life sciences research and drug discovery. In addition, we acquired Biometric Imaging Inc., whose cell analysis system for clinical applications complements our own flow cytometry capabilities, and Transduction Laboratories, which focuses on reagents for research for cell biology. We signed key agreements with Saf-T-Med, Inc., Med-Design Corporation and QIAGEN N.V. In the first two, we now have acquired technology and exclusive rights to spring-based retracting needle technology that will be major enhancements to our burgeoning safety-engineered product portfolio. With QIAGEN, we formed a worldwide joint venture for the collection and processing of nucleic acid samples. New product development As a result of our concerted and ongoing efforts, BD now has the richest and most promising new product pipeline in its history. Highlights of the year included the introduction of our BDProbeTec ET molecular diagnostic instrument outside the United States, our first BD.id implementations and, of greatest significance, the expansion of our new generation of advanced protection devices. Geographic expansion The year was one of consolidation, however, we did open our new plant in India, the largest facility of its type in Asia, with a capacity to produce well over a billion disposable needles and syringes annually. As Asian economies recover, we expect stronger revenues to follow. We have implemented a smooth transition of Boin Medica Co., Ltd., South Koreas largest medical supplies company, after acquiring it in fiscal 1998, and we expect it to support our future growth in South Korea and neighboring countries. In addition, we made progress with our presence and infrastructure in Latin America, Eastern Europe and the Middle East.
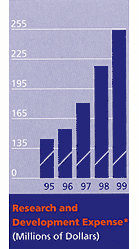
*In-process research and development charges of $49 million, $30 million, and $15 million associated with acquisitions were recorded in 1999, 1998, and 1997, respectively. |
||||
|